Step by step western blot procedure7/27/2023 S-S disulfide bonds to SH and SH) and thus allows separation of proteins by their molecular weight. SDS-PAGE (SDS polyacrylamide gel electrophoresis) maintains polypeptides in a denatured state once they have been treated with strong reducing agents to remove secondary and tertiary structure (e.g. The nature of the separation depends on the treatment of the sample and the nature of the gel.īy far the most common type of gel electrophoresis employs polyacrylamide gels and buffers loaded with sodium dodecyl sulfate (SDS). Separation of proteins may be by isoelectric point (pI), molecular weight, electric charge, or a combination of these factors. The proteins of the sample are separated using gel electrophoresis. Protease and phosphatase inhibitors are often added to prevent the digestion of the sample by its own enzymes.Ī combination of biochemical and mechanical techniques – including various types of filtration and centrifugation – can be used to separate different cell compartments and organelles. However, it should be noted that bacteria, virus or environmental samples can be the source of protein and thus western blotting is not restricted to cellular studies only.Īssorted detergents, salts, and buffers may be employed to encourage lysis of cells and to solubilize proteins. Cells may also be broken open by one of the above mechanical methods. In most cases, solid tissues are first broken down mechanically using a blender (for larger sample volumes), using a homogenizer (smaller volumes), or by sonication. Samples may be taken from whole tissue or from cell culture. Steps in a western blot Tissue preparation Detection of RNA is termed northern blotting. Neal Burnette and is a play on the name Southern blot, a technique for DNA detection developed earlier by Edwin Southern. The name western blot was given to the technique by W. The method originated from the laboratory of George Stark at Stanford. Other related techniques include using antibodies to detect proteins in tissues and cells by immunostaining and enzyme-linked immunosorbent assay ( ELISA). This method is used in the fields of molecular biology, biochemistry, immunogenetics and other molecular biology disciplines. 
Commercial antibodies can be expensive, though the unbound antibody can be reused between experiments. There are now many reagent companies that specialise in providing antibodies (both monoclonal and polyclonal antibodies) against many thousands of different proteins. The proteins are then transferred to a membrane (typically nitrocellulose or PVDF), where they are probed (detected) using antibodies specific to the target protein. It uses gel electrophoresis to separate native or denatured proteins by the length of the polypeptide (denaturing conditions) (Figure 1) or by the 3-D structure of the protein (native/ non-denaturing conditions). Our featured application notes below provide further details.The western blot (alternately, immunoblot) is a method to detect a specific protein in a given sample of tissue homogenate or extract. Utilizing this substrate-free assay, researchers can achieve equivalent sensitivity to traditional chemiluminescence assays while consolidating western blot, ELISA, and other applications onto a single reader. Each type of detection has advantages and disadvantages that need to be considered when selecting a method.Īnother method that we have introduced the ScanLater™ Western Blot Detection System, enables first-of-its-kind western blot detection in a multi-mode microplate reader platform. For example chemiluminescence can be detected using x-ray film or digital imaging equipment while a fluorescent secondary antibody requires a fluorescence imager. These methods are described below and require different equipment for detection. There are several western blot methods to consider depending upon the secondary antibody used, detection of the target protein may be colorimetric, chemiluminescent, or fluorescent. 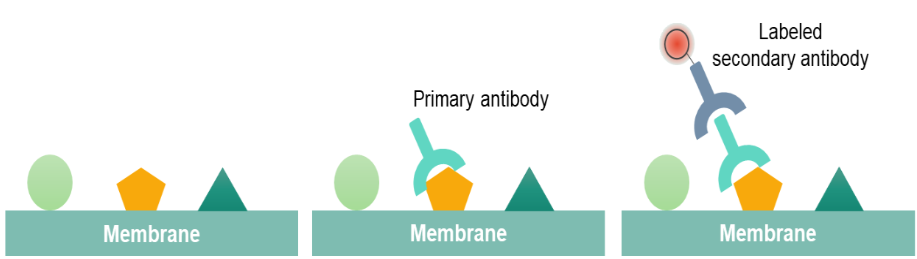
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
